

Custom Search
|
|

|
||
 MIL-HDBK-419A
7 . 8 BOND CORROSION. Corrosion is the deterioration of a substance (usually a metal) because of a reaction
with its environment. Most environments are corrosive to some degree. Those containing salt sprays and
industrial contaminants are particularly destructive. Bonds exposed to these and other environments must be
protected to prevent deterioration of the bonding surfaces to the point where the required low resistance
connection is destroyed.
7.8.1 Chemical Basis of Corrosion.
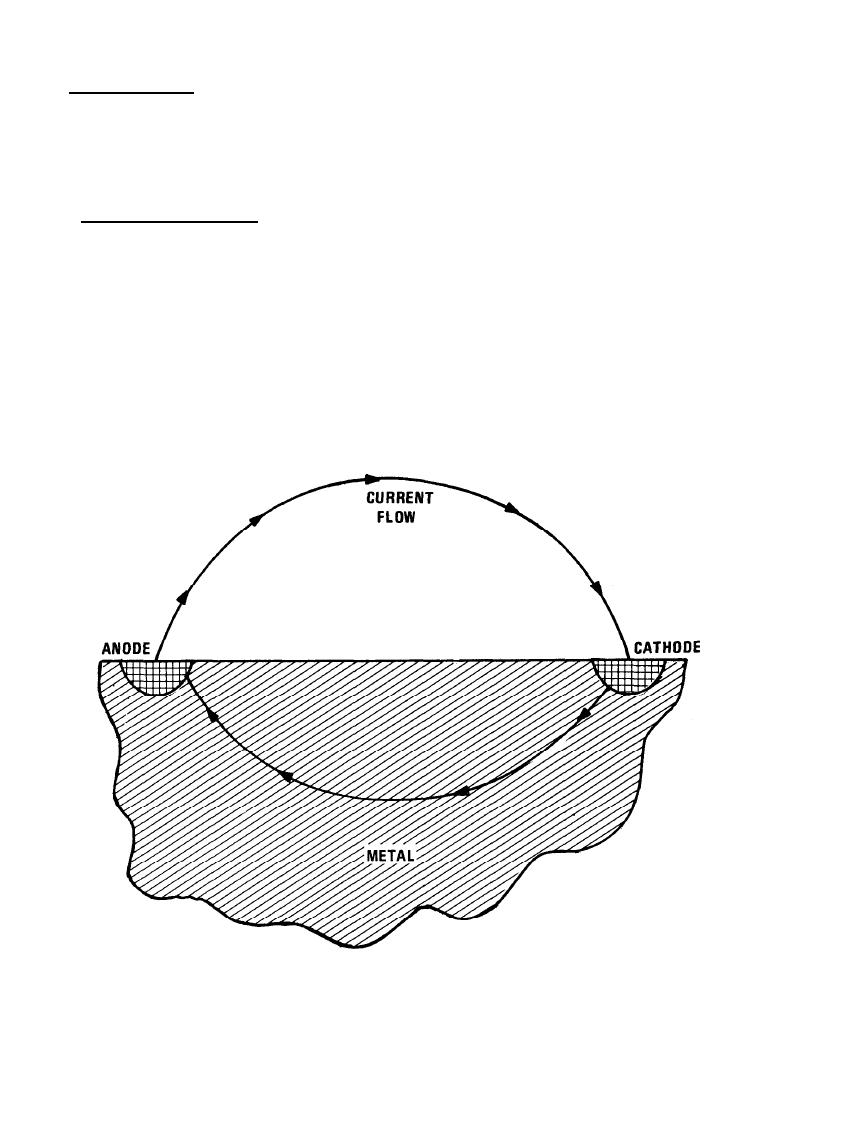
The basic diagram of the corrosion process for metals is shown in Figure 7-17. The requirements for this
process to take place are that (1) an anode and a cathode must be present to form an electrochemical cell and
(2) a complete path for the flow of direct current must exist. These conditions occur readily in many
environments. On the surface of a single piece of metal, anodic and cathodic regions are present because of
impurities, grain boundaries and grain orientations, or localized stresses. These anodic and cathodic regions are
in electrical contact through the body of metal. The presence of an electrolyte or conducting fluid completes
the circuit and allows the current to flow from the anode to the cathode of the cell.
ENVIRONMENT
(ELECTROLYTE)
Figure 7-17. Basic Diagram of the Corrosion Process
7-30
|
 
|
|
 |
||